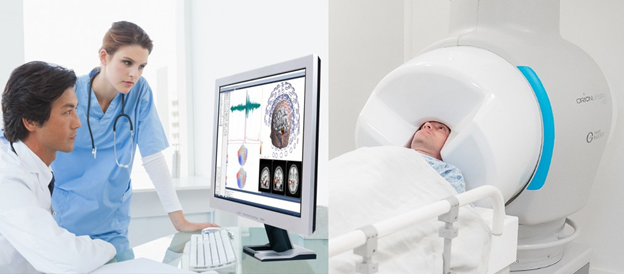
Perhaps now at the nascent stage which magnetic resonance imaging was 40 years ago, the development of magnetoencephalography could be headed for a similar path of growing adoption in the medical community. MEG is a neuroimaging technique for mapping brain activity by recording magnetic fields produced by electrical currents occurring naturally in the brain.
One company at the forefront of the pioneering development is Compumedics Neuroscan, whose Orion LifeSpan system, shown above, has been gaining acceptance and regulatory approval. Last year it received clearance from the US Food & Drug Administration for its Orion LifeSpan single Dewar MEG system, opening the door for routine clinical use of the device, primarily for epilepsy and pre-surgical brain function mapping. The clearance unlocks another important aspect, that sites using the device may now bill both private and public insurance plans for MEG examinations, an important consideration for healthcare facilities when purchasing medical equipment.

“Compumedics Neuroscan is very pleased to have achieved this important milestone for the Orion LifeSpan MEG,” said Dr. David Burton, chairman and CEO of Compumedics. “Whilst expected, it is nevertheless satisfying and validating after all the R&D over many years. Much like MRI technology in the late 1980s which transitioned from research to clinical application, we expect MEG to follow a similar trajectory.”
“In addition to the technical accomplishments achieved by the company and our partners at the Korea Research Institute of Standards and Science (KRISS), it represents a foundation for the commercialization of our MEG technology,” he added. “This market clearance will allow us to transition from product development to full commercialization. Compumedics continues to actively pursue 40 identified MEG opportunities around the world, with four opportunities substantively advanced.”
In the late 1980s there were about 200 or so MRI systems installed, like MEG today. These were largely used for research at that time before billable codes became available in the 1990’s for MRI. Today, about 36,000 MRIs installed globally with 2,000 to 3,000 systems sold annually.
With the FDA approval in place, Compumedics may now enter the lucrative US clinical market with its single Dewar MEG technology. Each system will typically sell for around $3-4 million, depending on specification.
A dual adult/child Dewar is also being developed by the company. While its first commercial installation of the dual system has been delayed during the Covid-19 pandemic, it has been rescheduled for sometime in 2021, the company reported in February. It is destined for the prestigious Barrow Neurological Institute in Phoenix, Arizona, where a single-Dewar system is already in place. A Dewar is a specialized vacuum flask for storing cryogenic material, necessary to keep the system at low temperature.
At the heart of the system is a patented Double Relaxation Oscillation Superconducting Quantum Interference Device (DROS SQUID) sensor, providing advanced MEG sensitivity and spatial resolution. The technology has evolved from more than thirty years of experience with MEG and EEG technologies, says Compumedics, employing highly sensitive magnetic field detectors and low-noise amplifier electronics, which have been developed at both the Korea Research Institute of Standards & Science (KRISS) and within Compumedics Neuroscan, says the company. In 2016, the two entities negotiated a 20-year exclusive technology transfer and license agreement to produce the Orion LifeSpan.
Based in Victoria, Australia, Compumedics is a medical device company that specializes in diagnostic technology for sleep, brain and ultrasonic blood-flow monitoring applications. The company owns US based Neuroscan and Germany based DWL Elektronishe GmbH. It was founded by Burton in 1987, the same year it designed and installed the first Australian, fully computerized sleep clinic at Epworth Hospital in Melbourne, and followed with developing products for sleep clinic and home monitoring markets. For more info, see www.compumedics.com.