
Fresh from success battling Covid-19, Australian biotech company AnteoTech is setting up for rapid expansion based upon its AnteoBind technology for improved molecular conjugation. Armed with $12 million in new funding, the company is developing more tests and its own line of test equipment for life sciences markets.
AnteoTech offer a range of ready-to-use kits based on its technology to streamline and improve the conjugation process. The versatile kits are compatible with carboxylated magnetic particles, latex and europium nanoparticles. Among its conjugation kits and reagents, the company offers coupling kits of 200 nm magnetic particles and 1µm magnetic particles for magnetic particle-based assay development & bio-separation.

Funds from the recent stock placement, announced April 28, will be used to scale up the roll-out of the company’s EuGeni reader and invitro rapid diagnostic test used in Covid-19 detection, also to accelerate its pipeline of other assay tests. “The support for this placement is a strong endorsement of AnteoTech’s growth strategy across both its Life Sciences and Energy divisions. Both business units have technology solutions that are applicable to large and deep global markets,” said Derek Thomson, CEO. Positioned as a surface management technology company, AnteoTech, which is based in Brisbane, also enables improved capabilities with lithium materials for the battery industry, another area in which it is targeting rapid growth.

EuGeni is a fast, accurate and compact instrument for rapid point-of-care testing. With a small and portable benchtop design, it can be easily transported in the clinic and on the field for use by healthcare professionals. Compatible with magnetic particle assays, the rapid test platform provides the basis of an entirely new potential revenue stream for the company. Business growth is expected with the addition of not only the proposed COVID-19 suite of tests but also the development of a range of new diagnostic solutions in the areas of infectious, bacterial and viral conditions, said Thomson.
Its seven-year collaboration with Australian diagnostics company Ellume has led to much of AnteoTech’s recent success. Ellume uses AnteoBind technology to support significantly improved sensitivity in a number of tests, including its COVID-19 Home Test which does not require sending samples to a lab, similar to how home pregnancy tests work. In February, the U.S. government pledged $230 million to Ellume to ramp up production of the over-the-counter test.
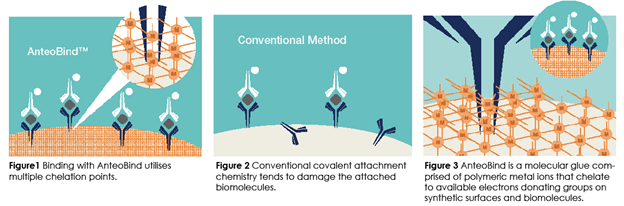
AnteoBind technology
AnteoBind is an aqueous, nanosized molecular glue made that is used in a rapid, single-step process to facilitate multivalent coordination to bind carbohydrate polymers, antibodies, and proteins to synthetic substrates in assay development. A single binding point would not be strong enough to bind a metal ion to a synthetic surface or a biomolecule to the metal ion. AnteoBind overcomes this limitation by using polymeric metal ions of Europium that form multiple binding points with both the underlying surface and the biomolecule of interest. For more info, see www.anteotech.com and www.ellume.com.



