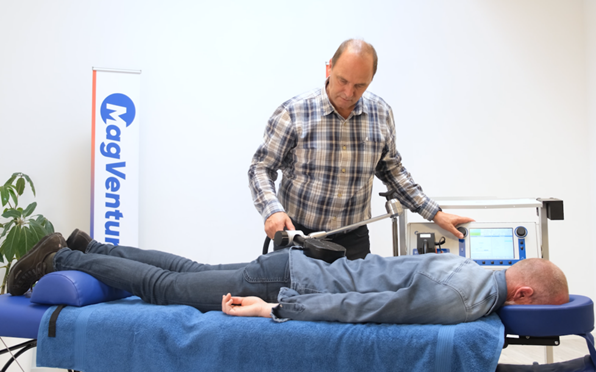
MagVenture, celebrates a new milestone with the launch of MagVenture Pain Therapy—an FDA-cleared solution for chronic pain relief. For more than three decades, the Danish company has been at the forefront of using magnetic stimulation to enhance the lives of patients suffering from depression and other psychiatric disorders using its transcranial magnetic stimulation (TMS) systems. Now, with the introduction of MagVenture Pain Therapy, it is extending the technology to bring those solutions to sufferers of chronic pain.
“Today, we take all our experience and commitment to patient care to new heights. We’re excited about the possibilities of expanding our technology to reach individuals who are living with chronic pain, offering them our perspective on pain management”, says Kerry Rome, senior vice president of sales at MagVenture Inc.
The system of a specialized coil and a stimulator employs powerful focused magnetic pulses directed near the injured nerve, providing a non-invasive, painless approach to stimulate peripheral nerves for clinically proven pain relief. Unlike traditional treatments for chronic nerve pain, MagVenture Pain Therapy offers a unique neuromodulation technique.
What sets the therapy apart is its versatility and ease of use. Patients can experience relief with sessions lasting just 13 minutes, spread over a two-month period. The non-pharmacologic treatment is a significant advancement for those seeking a safe, efficient alternative to surgery or medication for chronic pain relief.

Privately-owned and headquartered in Denmark, MagVenture has been pioneering cutting edge TMS solutions for more than 30 years. Its TMS systems are used for a wide range of applications in both research and treatment in the fields of psychiatry, neurophysiology, neurology, cognitive neuroscience, and rehabilitation.

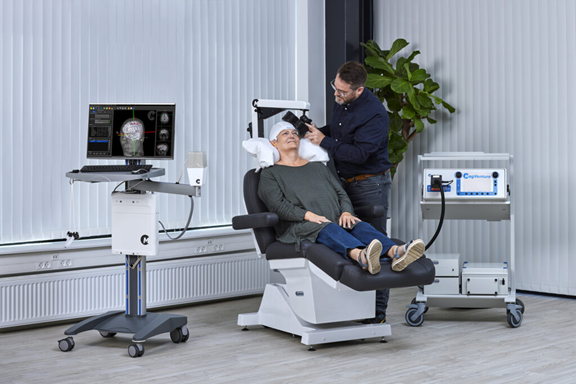
In another recent clearance, last year the company received FDA approval for use of its TMS Atlas Neuro Navigation System for high-precision, personalized TMS coil positioning. Atlas uses the patient’s unique magnetic resonance imaging data set for precise, individualized coil positioning to target the exact TMS treatment spots in the brain. By using electromagnetic tracking which eliminates line-of-sight issues, Atlas gives the operator greater freedom of movement during TMS treatments. “While the existing MagVenture marking plate system is a reliable method of coil positioning, our Atlas Neuro Navigation System offers a more streamlined and sophisticated experience which may contribute to increased confidence in the procedure,” said Rome. For more info, see www.magventure.com.



