
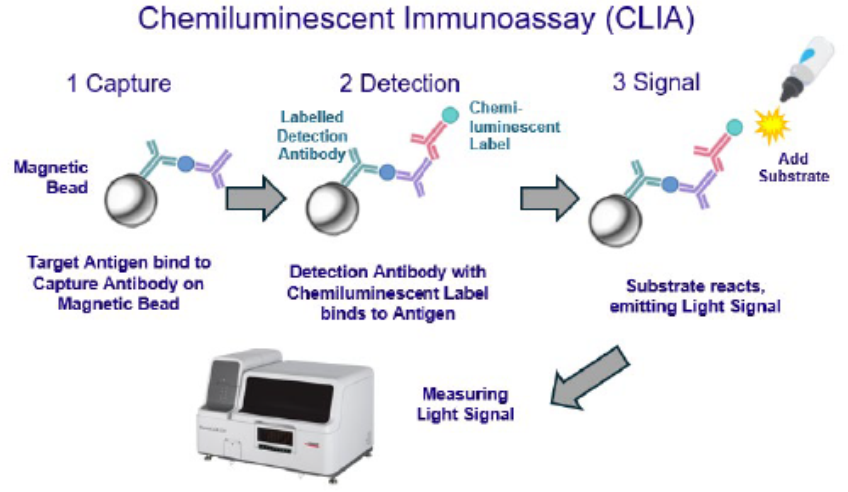
Intent on expanding into major growth markets and with richer solutions, AnteoTech has begun a new collaborative immunoassay product development program with its AnteoBind NXT activated magnetic particles for use in chemiluminescent immunoassays (CLIA). Based in Brisbane, Australia, the firm is working with a large but unnamed global life sciences company to develop an activated particle product for use in the global CLIA market.
The new product is intended to address the CLIA market’s growing need for highly sensitive, specific, reproducible, cost effective and easy to use magnetic particles. These particles are central to high throughput, fully automated CLIA platforms used in today’s clinical laboratories. Subject to successful test work, AnteoBind NXT could be supplied as a particle activator within CLIA kits used across this growing global market, valued at US$13 billion in 2024.
AnteoTech will receive A$64,500 for services and materials under a 50:50 shared cost contract for the Initial Work Program, which is scheduled for completion by the end of FY 2026. Successful completion is expected to lead towards a joint product development agreement in relation to CLIA market entry.
In-house testing of magnetic particles using AnteoBind NXT, compared to Tosyl chemistry, which is the most widely used single step coupling chemistry in CLIA, has demonstrated significant performance and cost benefits, including up to six times lower antibody usage. Capture antibodies can account for up to 35% of total assay costs.
The program follows proactive re-engagement with a number of large global life sciences companies, supporting AnteoTech’s strategy of taking AnteoBind NXT from a product improvement additive to a core ingredient in performance leading, low-cost products in the life sciences sector, underpinning additional sales growth and value creation, essentially moving it up the value chain.
Basically, AnteoTech is no longer content to supply additives to other people’s tests; it’s now eyeing a central role in two of the biggest immunoassay formats on the market – ELISA (enzyme-linked immunosorbent assays) and CLIA.




